 
The final data column compares this percentage against the percentage of all minerals that contain the element listed in each row. A special application of cesium is that it is used in the manufacturing of most accurate atomic clock. Element: Cesium Symbol: Cs Atomic Mass: 132. Atomic number: 55: Relative atomic mass: 132.905: State at 20☌: Solid: Key isotopes: 133 Cs: Electron configuration Xe 6s 1: CAS number: : ChemSpider ID. 
The second data column lists this number as a percentage of all minerals listed with Caesium. Element: Chlorine Symbol: Cl Atomic Mass: 35.453 of Atoms: 1 Mass Percent: 21.058. The first data column contains the total number of minerals listed with Caesium and the element listed for that row. Using only the information provided in your question, one-twelfth the mass of an atom of C-12 is 1 unified atomic mass unit or 1 u, so the mass of a Cs atom. Atomic Number: 55 Symbol: Cs Atomic Weight: 132. The isotope Cs-137 is used in cancer treatments, to irradiate foods, and as a tracer for drilling fluids in the petroleum industry. A 'cesium(-beam) atomic clock' (or 'cesium-beam frequency standard') is a device that uses as a reference the exact frequency of the microwave spectral line emitted by atoms of the metallic element cesium, in particular its isotope of atomic weight 133 ('Cs-133'). Note that unlike other sections on this page this includes non-essential elements. Cesium is used in atomic clocks, photoelectric cells, as a catalyst to hydrogenate organic compounds, and as a getter in vacuum tubes. Atomic Structure of Cesium Atomic Radius: 3.34 Atomic Volume: 71.07cm3/mol Covalent Radius: 2.35 Cross Section (Thermal Neutron Capture)a/barns: 29. This table compares the known valid mineral species listed listed with Caesium and the other elements listed based on the official IMA formula. 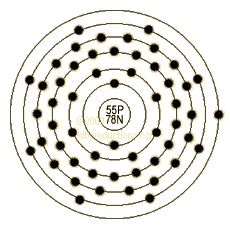
Minerals where Cs was previously thought to be a colouring agent probably contain other ions such as Mn#2+#.Ģ7 valid species containing essential CaesiumĬs + enters later phases in igneous rocks due to its large size.Ītom mole fraction relative to Si=1 (% uncertainty)Įlement association of Caesium in the Mineral World It is a very ductile, pale metal, which darkens in the presence of trace amounts of oxygen. Wikipedia WebElements Los Alamos National Laboratory Theodore Gray's Ĭaesium as a chromophore in minerals and gemsĭoes NOT cause colour as it has no electrons in d-orbitals. Physical properties Of all elements that are solid at room temperature, caesium is the softest: it has a hardness of 0.2 Mohs. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
